MetrumRG is proud to continue to be an educational resource for our community by offering open, free pharmacometrics training through a variety of mediums. The range of courses and training content, listed here: metrumrg.com/courses/, includes a series of courses that were intended to progress from introductory through more advanced topics according to "MI" course number, e.g., MI210 (intro to pharmacometrics modeling and simulation) would be completed prior to MI212 (intermediate/advance topics), etc.
We have since added several "special topic" courses. In general, these are likely best done after completing the "MI" courses.
We also encourage individuals to periodically check our github site (github.com/metrumresearchgroup) -- we often post our workshop materials to this site. For example, the hand-outs/slides for the two workshops we taught at ACoP10 (Fall 2019) have been posted here:
Expand your knowledge of quantitative pharmacology--check out our online training resources and newly launched discussion forum!
Topics: Metrum News, Open Science
MetrumRG "COOPED UP" Webinar Series to Begin
Metrum Research Group's Cellar Office OPen-EDUcation in Pharmacometrics (“COOPED UP”) webinar series will be kicking off next month! These webinars will feature a range of topics, discussed in vignette formats, to share with our community the same training and development in which our staff routinely engages.
Topics: Open Science
Showing Stripes and more for Rare Disease Day 2020
Topics: Community Outreach
In my first post in this series, I proposed a “starter kit” for developing a statistical argument. This “starter kit” included considerations related to both the direction of effects (“[Is] the association directionally consistent with known mechanistic accounts (e.g. based on enzymology)?”) and the magnitude of effects (“Does the association have a magnitude that is consequential in some practical sense?”). In this post, I argue that “significance language” can play a valuable role in discussing the estimated direction of effects, even if supplementary approaches are needed to address more complex questions about magnitude.
Topics: Scientific Writing, Significance, Confidence Intervals, P-values
Statistical Significance is a Result, Not a Conclusion
In my previous post, I proposed a “starter kit” for developing a statistical argument. An essential ingredient in this “starter kit” was the identification of the context of inquiry as being confirmatory, explanatory, or something in between. In this post, I propose several criteria for defining a confirmatory context, and I suggest principles for using the language of “statistical significance” when confirmatory contexts do not obtain.
Read the latest news, events, and publications from Metrum Research Group in our Winter 2020 Newsletter.
Here’s what you will find in our newsletter:
- Join MetrumRG at ASCPT: Our workshop, our Booth and an award!
- New models and blog posts on our website!
- Announcement about our newest team members!
- Upcoming events our scientists will be attending or presenting at.
- Publications and presentations by MetrumRG scientists.
Topics: Metrum News
A Significant Tilling of the Statistical Soil: The ASA Statement on Statistical Significance and P-values
This is the first in a series of blog posts that will focus on practical problems of summarizing statistical evidence in the context of biomedical science, using the 2016 ASA statement on p-values and significance as a guide. I hope and expect that these posts will find a readership that does not entirely agree with my views in all of their particulars, and that this readership takes advantage of the blog / combox format to register both their agreements and disagreements. Merely good ideas are insufficient to the task at hand ; a shared understanding has to develop in the community of practice ; my hope here is to provide a forum for discussion for that community.
Topics: Statistics, Scientific Writing, Significance, Confidence Intervals, P-values
Check out these recent publications by authors at Metrum Research Group and our collaborators!
Topics: Metrum News
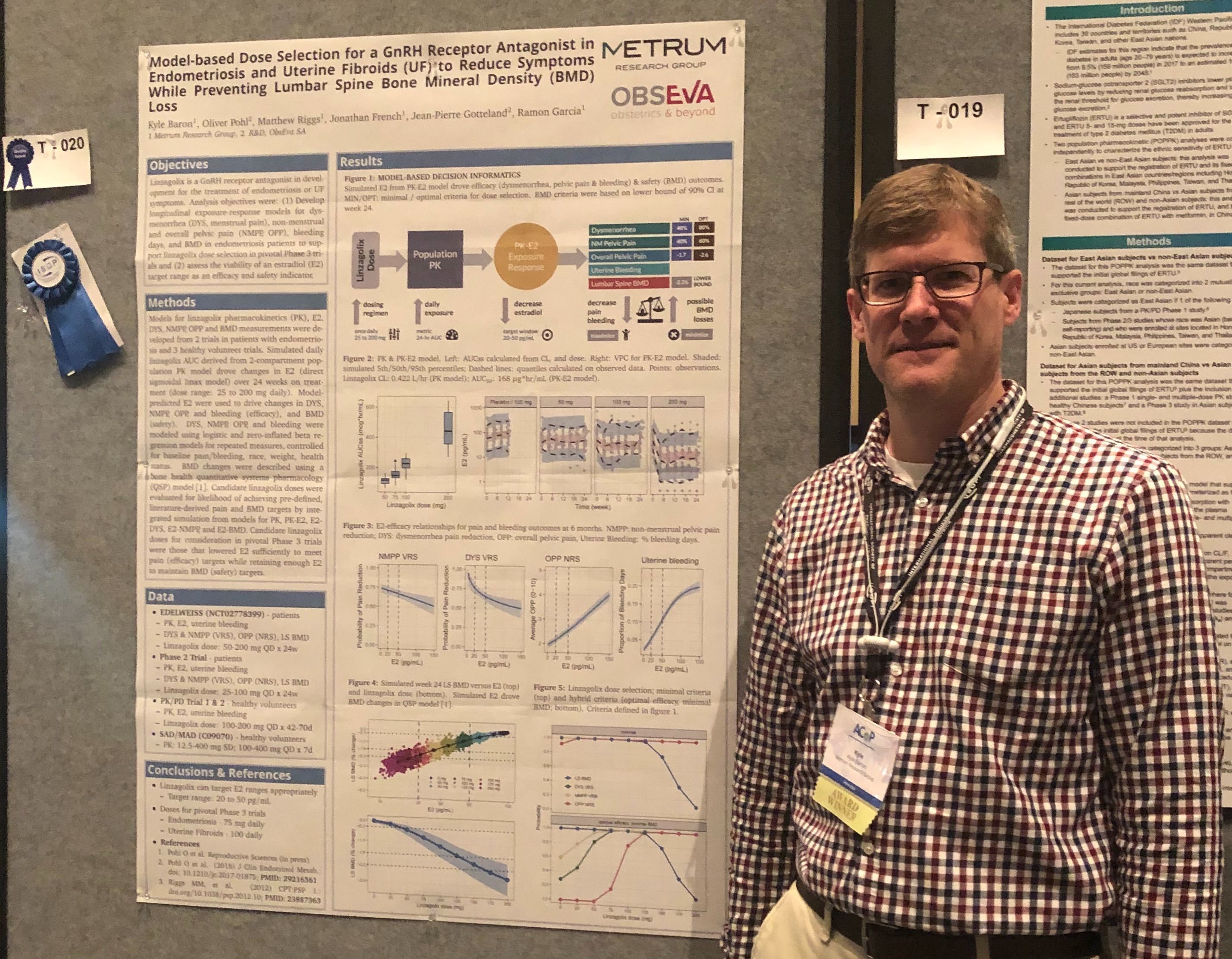
MetrumRG scientists traveled to Orlando, FL to join the American Conference on Pharmacometrics for their 2019 annual meeting in October.
Topics: Metrum News




.jpg)